MDMA vs MDA vs MDEA: Differences & Therapeutic Use in Psychedelic Therapy Training 2026
MDAM Crystal
In the rapidly growing field of psychedelic-assisted therapy, entactogens like MDMA (3,4-methylenedioxymethamphetamine), MDA (3,4-methylenedioxyamphetamine), and MDEA (3,4-methylenedioxy-N-ethylamphetamine, also known as MDE or "Eve") foster emotional openness, empathy, and introspection. These qualities offer potential for mental health professionals treating trauma, PTSD, anxiety, and relational challenges.
At Changa Institute, we provide accredited psychedelic therapy training and certification programs for licensed mental health practitioners, wellness professionals, and facilitators. Our evidence-based curricula equip you to guide clients ethically through preparation, supervised sessions, and integration. This article offers an overview of MDMA, MDA, and MDEA differences, MDMA's therapeutic protocols and breakthrough results, and why specialized training is vital for safe, effective practice.
All discussions are educational, MDMA, MDA, and MDEA remain Schedule I substances in most jurisdictions. Use is limited to approved clinical trials, licensed settings (e.g., Oregon/Colorado psilocybin programs), or future regulated frameworks under supervision.
Are MDMA, MDA, and MDEA Entactogens?
Yes, all three are entactogens (or empathogens), a term coined by David Nichols in 1986 for compounds enabling "touching within": self-awareness, emotional processing, trust, and connection without dominant hallucinations source.
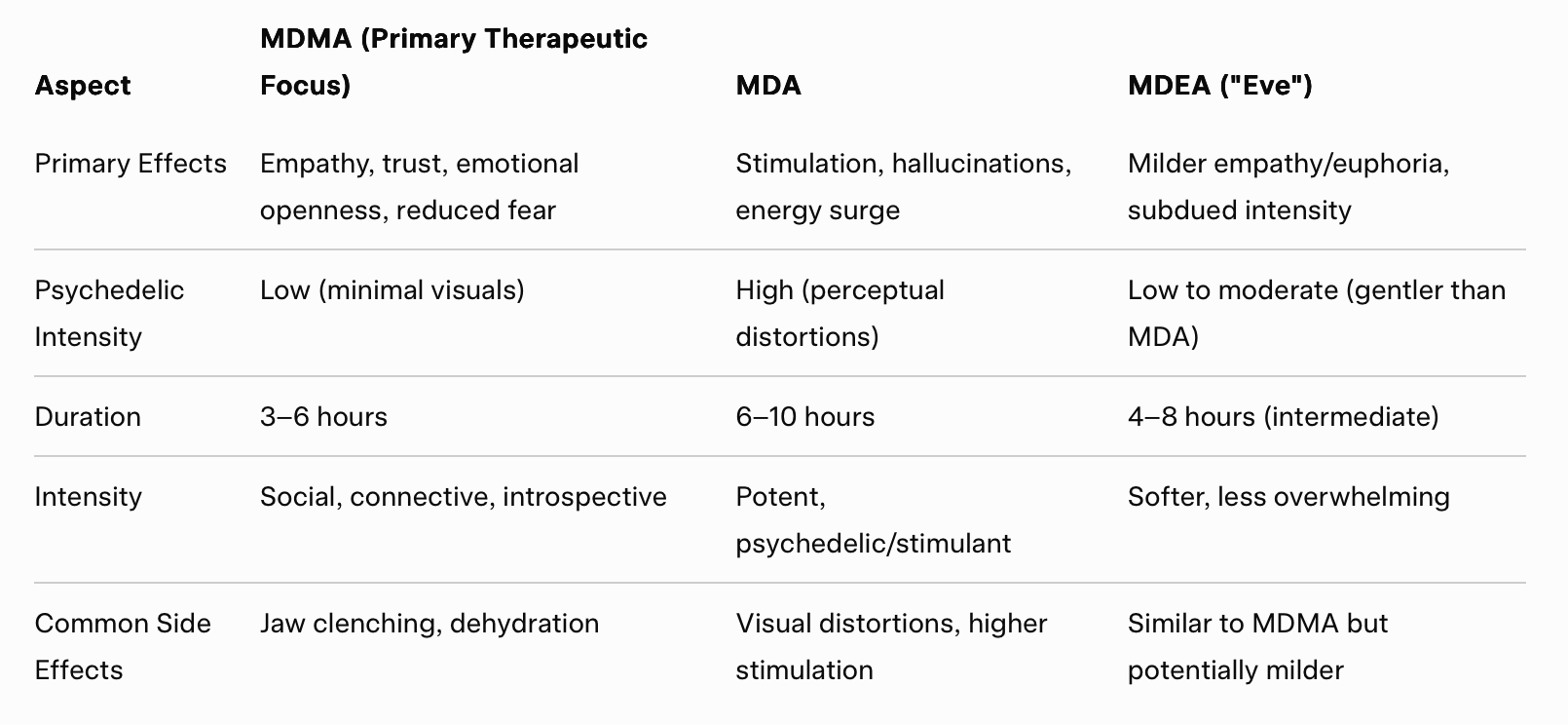
MDMA excels therapeutically due to its serotonin-dominant profile. MDA (structural parent) adds stronger dopamine-driven stimulation and visuals. MDEA (N-ethyl analog) offers a milder, intermediate profile, less intense than MDMA and far less psychedelic than MDA.
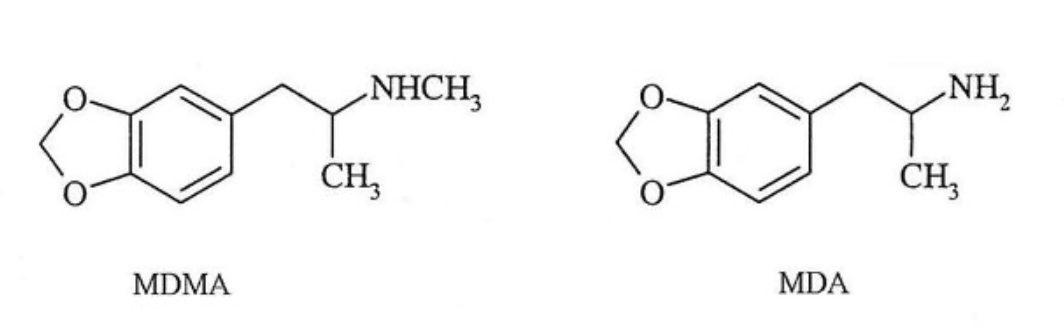
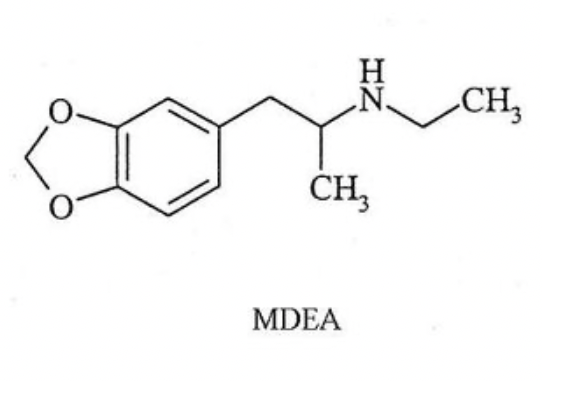
Chemical and Structural Differences
Progressive N-alkylation shifts effects: MDA → more hallucinogenic/stimulant; MDMA → optimal empathy balance; MDEA → softer/milder pharmacology review.
These are amphetamine derivatives with a methylenedioxy ring:
MDA: Primary amine (no N-alkyl group).
MDMA: N-methyl group.
MDEA: N-ethyl group.
Effects Comparison
A 2025 crossover study confirmed MDA's longer duration (6.1 h vs. MDMA's 4.1 h) and stronger perceptual alterations compared to MDMA study; MDEA data is more limited (mostly older reports showing a "gentler" profile).
Risks and Safety in Therapeutic Contexts
All three compounds carry risks, including serotonin syndrome, hyperthermia, and potential neurotoxicity, particularly with repeated or high-dose use. However, in controlled, low-dose therapeutic settings, MDMA demonstrates a favorable safety profile, with most adverse events being transient and mild-to-moderate.
The most common side effects reported in Phase 3 MDMA-assisted therapy trials include:
Jaw clenching / bruxism
Nausea
Decreased appetite
Dry mouth
Sweating
Anxiety or mild panic (usually early in the session)
Temporary fatigue or low mood in the days following ("come-down")
These effects are typically short-lived (hours to a few days) and manageable with supportive care, hydration, and integration support. MDA tends to produce more intense stimulation and perceptual changes, increasing the likelihood of discomfort, while MDEA appears milder in limited reports but lacks comparable clinical data.
Ethical practice requires thorough client screening (e.g., excluding those with cardiovascular issues), informed consent, harm reduction strategies, and trained facilitation, all core competencies covered in Changa Institute's psychedelic-assisted therapy certification programs.
How MDMA Is Used in Therapy: Protocols and Breakthrough Results
MDMA-assisted therapy uses structured protocols (e.g., MAPS/Lykos models):
Preparation (non-drug sessions): Rapport, intentions, fear processing.
Dosing Sessions (2–3): 80–120 mg MDMA (optional booster), non-directive therapist support in safe set/setting.
Integration (post-session): Insight processing, change consolidation.
Breakthrough Phase 3 results (MAPP1/MAPP2 trials):
67–71% of PTSD participants no longer met criteria (vs. ~30% placebo) Nature Medicine 2023; MAPP2 details.
~46% full remission in key studies.
Sustained benefits: Symptom reduction, improved relationships/quality of life (months to years).
Strong for treatment-resistant PTSD (e.g., veterans); emerging promise in couples therapy for relational trauma, boosting empathy, communication, bonding.
MDA and MDEA have negligible therapeutic research; MDMA's evidence base drives its lead.
Current Research Landscape (February 2026)
MDMA dominates; MDA/MDEA are comparators with limited momentum. No FDA approval—Lykos pursues resubmission post-2024 CRL via additional Phase 3 and data review Lykos update.
Key players:
PharmAla Biotech — Supplies LaNeo™ MDMA for trials (e.g., recent Østfold Hospital MDD study) press release.
Lykos Therapeutics — Advances MDMA protocols for PTSD/anxiety.
MAPS–Columbia Partnership — Couples/relational focus.
Tactogen — Preclinical novel entactogen analogs (e.g., TACT411 series) for safer profiles; holds key patent (US 11767305, 2023) Tactogen announcement.
Global guidelines (e.g., Australia's NHMRC framework for MDMA-AP in PTSD) support expansion NHMRC guideline; economic models indicate significant cost savings in trauma care.
Why Choose Changa Institute for Psychedelic Therapy Training and Certification?
As regulations evolve, practitioners need rigorous skills for ethical facilitation. Changa Institute offers state-approved programs (e.g., Oregon OHA/HECC, Colorado DORA) in psilocybin facilitation, psychedelic integration, and broader psychedelic-assisted therapy training, covering ethics, trauma-informed care, session support, and integration.
Our certification empowers you to:
Guide evidence-based experiences ethically.
Meet licensure requirements.
Support trauma healing, relational growth, and wellness.
Ready to advance your practice? Explore our psychedelic therapy certification programs or contact hello@changainstitute.com for upcoming cohorts.